Manufacturing Execution System
Built on a Decade of Innovation. Designed for the Next One.
Schedule a DemoThis Is Not Your Legacy MES
Operator-Owned. Not IT-Dependent.
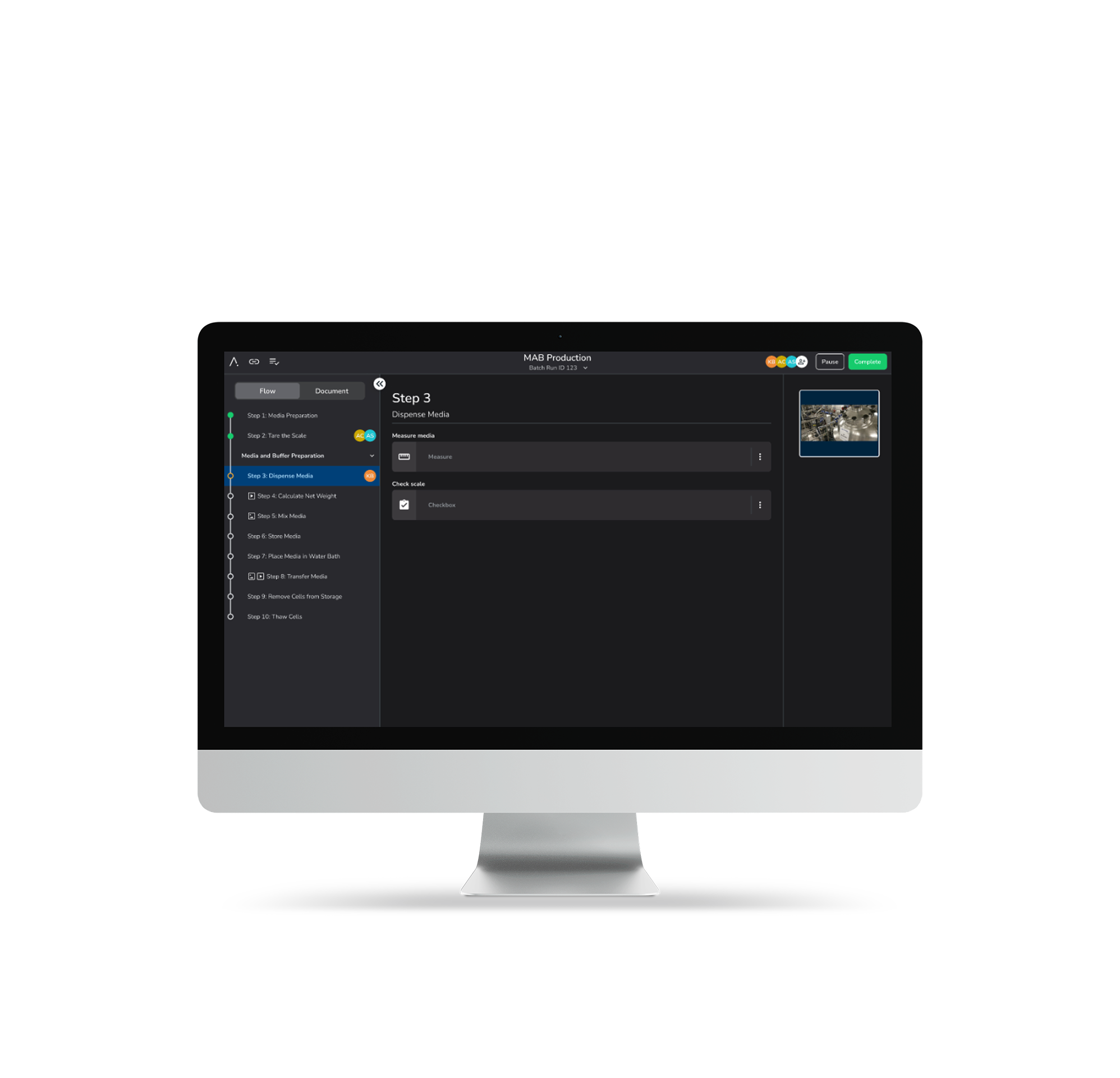
The people who know the process should own the process. SMEs author, modify, and optimize recipes directly through no-code Designer Mode — no IT tickets, no developer queues, no waiting.
Dynamic Execution. Not Rigid Scripts.
Ad hoc recipe changes mid-run. Dynamic parameter updates. Batch-level redlining. Real manufacturing doesn't follow a script — your MES shouldn't either.
Deploy in Weeks. Not Years.
Go live in 90 days. Roll out six sites in nine months. Group-activate entire regions simultaneously. This isn't a multi-year IT initiative — it's manufacturing at the speed of your business.
What Makes This a True Next-Gen MES
Operator-Owned Recipe Development
The people who know the process should own the process. Empower SMEs to author, modify, and optimize recipes directly — no IT tickets, no developer queues, no six-month change cycles.
Designer Mode puts recipe authoring in the hands of the people closest to the process.
SMEs design and modify clinical recipes with in-suite changes and real-time feedback to the design team
No-code drag-and-drop authoring with parallelization, branching, and enforcement rules
Green field digital-first operations from day one — skip the paper-to-digital migration entirely
Build dynamic, parameterized templates that scale across products, modalities, and global sites
SMEs design and modify clinical recipes with in-suite changes and real-time feedback to the design team
No-code drag-and-drop authoring with parallelization, branching, and enforcement rules
Green field digital-first operations from day one — skip the paper-to-digital migration entirely
Build dynamic, parameterized templates that scale across products, modalities, and global sites
Dynamic Batch Execution
Manufacturing doesn't follow a script. Neither should your MES. Make ad hoc recipe changes, update parameters mid-run, and adapt in real time without stopping production.
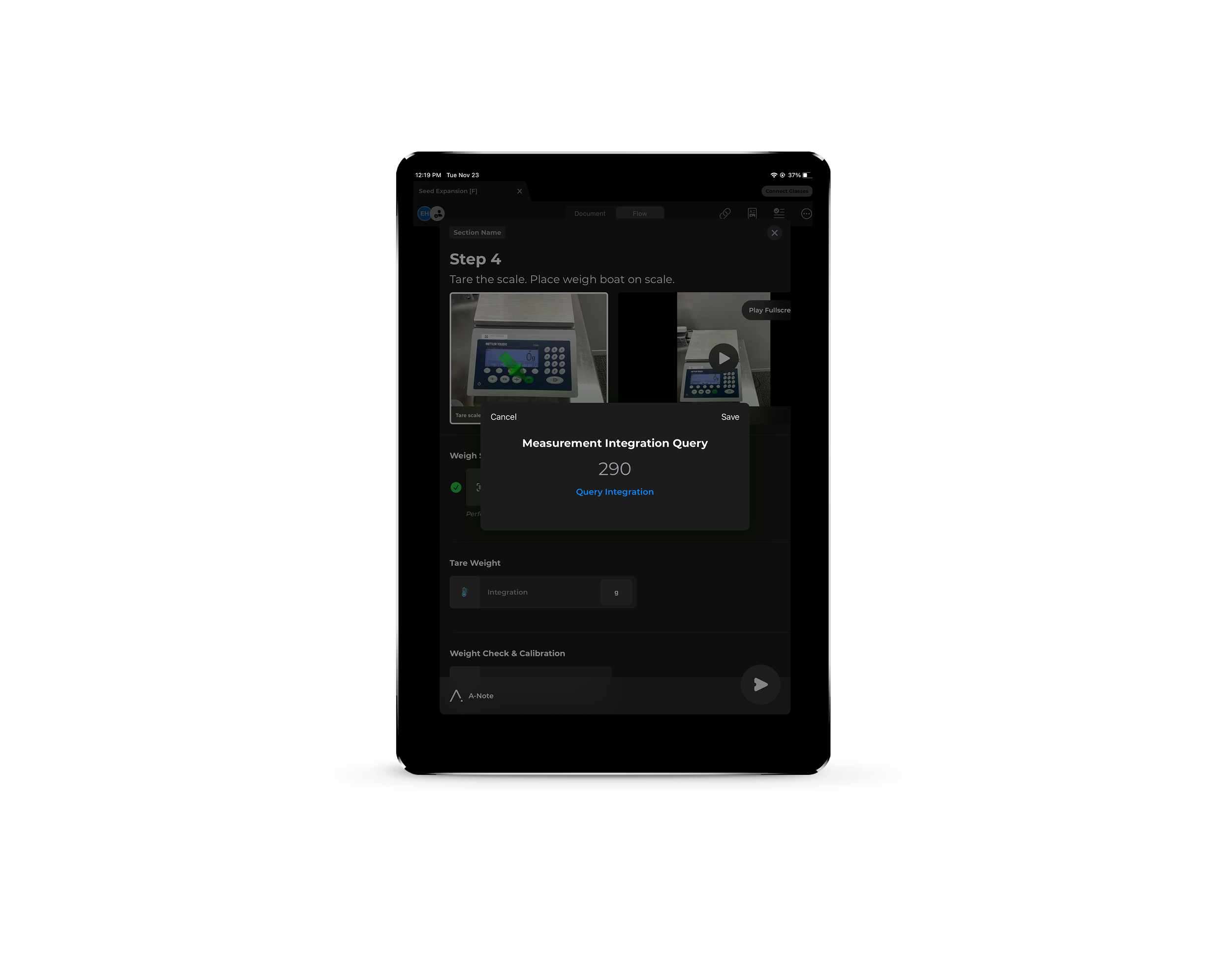
Batch Run Modification — ad hoc recipe changes, dynamic parameter updates, and mid-run iteration without halting production
Batch Parameter Groups — dynamic parameterization for multiple products through a single template, eliminating recipe sprawl
Redlining & N/A — capture on-the-fly clinical changes and mark steps not applicable in real time
Intuitive execution on any device — web, mobile, or hands-free — with automated data capture and built-in guardrails
Batch Parameter Groups — dynamic parameterization for multiple products through a single template, eliminating recipe sprawl
Redlining & N/A — capture on-the-fly clinical changes and mark steps not applicable in real time
Intuitive execution on any device — web, mobile, or hands-free — with automated data capture and built-in guardrails
Rapid Deployment at Enterprise Scale
Go live in 90 days. Roll out six sites in nine months. Group-activate entire regions simultaneously. This isn't a multi-year IT initiative — it's manufacturing at the speed of your business.
Select your first site, configure, validate, and manufacture — then group-activate remaining sites simultaneously with parallel configuration and PQ
Enterprise rollouts that used to take years now take months, with proven deployment playbooks across global pharma
Programmable guardrails and automated calculations reduce error at the source, while real-time exception notifications streamline review and release
Built-in QMS integration keeps deviation and CAPA management aligned and auditable across every site from day one
Enterprise rollouts that used to take years now take months, with proven deployment playbooks across global pharma
Programmable guardrails and automated calculations reduce error at the source, while real-time exception notifications streamline review and release
Built-in QMS integration keeps deviation and CAPA management aligned and auditable across every site from day one
Enterprise Network Operations
From external sponsor collaboration to global centers of excellence to one-click tech transfers — run your entire manufacturing network as one connected operation.
External Sponsor — work directly with clients and external teams through the platform with controlled access and real-time visibility
Global Center of Excellence — streamlined management of sites and teams, new platform validation, testing and UAT for the entire network
One-Click Tech Transfer — move records, recipes, and configurations across sites and teams instantly, eliminating months of manual rework
Site-specific customizations and mobile-ready access keep every team connected and compliant across your entire enterprise
Global Center of Excellence — streamlined management of sites and teams, new platform validation, testing and UAT for the entire network
One-Click Tech Transfer — move records, recipes, and configurations across sites and teams instantly, eliminating months of manual rework
Site-specific customizations and mobile-ready access keep every team connected and compliant across your entire enterprise
Built to Meet the Needs of Modern Manufacturing
Cloud-native
Manufacture on a reliable, scalable platform with access to seamless feature updates for continuous innovation
No-code
Accelerate adoption and unlock endless flexibility without the need for technical resources or coding experience
Operator Centric Design
Enhance the operator experience across mobile, web, and hands-free devices with a familiar modern tech feel
Secure, Compliant and CSA-ready
Built for global compliance, with ISO 27001, GDPR readiness, and full IQ/OQ validation for cGXP and 21 CFR Part 11.
Rapid Implementation
Deploy in 90 days or less to manufacture and realize value, faster