A1: The AI Agent for Manufacturing Teams
Respond to alarms, troubleshoot faster, eliminate paperwork, and prevent exceptions before they happen.
















An Agent for Every Role
A1's sub-agents automate the work you're spending hours on manually.
Things you can do today, but faster.
Workflows you never thought possible.
Apprentice 4.1 AI Model
General AI was built to talk – not to run production. We trained and constrained the Apprentice 4.1 model to outperform state-of-the-art LLMs in the dimensions that matter for manufacturing.
| Model | MFG Accuracy | MFG Specificity | MFG Relevance | Consistency | Compliance | Mean |
|---|---|---|---|---|---|---|
| Apprentice 4.1 | 4.8 | 4.9 | 4.933 | 5 | 5 | 4.927 |
| Claude Sonnet 4.6 | 4.6 | 4.8 | 4.633 | 4.7 | 5 | 4.747 |
| GPT-4o | 4.2 | 4 | 4.4 | 4.8 | 5 | 4.48 |
| GPT-4o Mini | 3.8 | 3.8 | 4.3 | 4.7 | 4.8 | 4.28 |
| Gemini 2.5 Flash | 3.5 | 3.4 | 3.4 | 3.9 | 4.1 | 3.66 |
One Model, Two Ways to Work
Use Apprentice 4.1 as a standalone AI agent or embedded within our Manufacturing Suite.
Pre-built Agents & Workflows ensure A1 can execute a repeatable, consistent and compliant workflow every time.
You can't afford to let AI make up a new workflow every time it runs. A1's Agent and Workflow capability allows you to define the exact steps and actions A1 should take every time it runs.
A1 creates production-ready artifacts — CAPAs, deviations, reports and more.
A1 doesn't just answer questions. It generates real manufacturing documents — from CAPA reports to process summaries — that you can review, edit, and share with your team.
A1 is not a chatbot. It proactively responds 24/7 to alarms, events, schedules, file changes and more.
Production doesn't wait for a prompt. A1 can be configured to proactively respond to 'triggers' so you don't have to constantly be in front of your computer - let A1 handle the noise so you can focus on what's important.
One Agent. Every System.
A1 Agents and Workflows execute across your entire manufacturing tech stack — ERP, MES, QMS, EAM, SCADA, Historian, PLCs, IoT, and flat files. Connect point-to-point or through a middleware with Webhooks, MQTT or OPC-UA.
A1 can communicate with you where you are - Email, SMS, WhatsApp, Slack, Teams, and more
When an issue happens on the floor, the last thing you want is to have to log in to an app. A1 can reach out to you on the channels you already use so you don't lose a second.
Built on the same infrastructure that has been powering our Manufacturing Suite for 12 years.
We've been deploying cloud-native manufacturing software for over a decade in the most difficult and regulated industries in the world. Designed to be fault-tolerant, geographically redundant, and always available, you can have the confidence to use A1 on your most critical workflows.
Free for individuals. Scale to your team, site and enterprise.
Use A1 for free to automate your manual work. When you're ready to scale, pick a plan that fits your operations and grow from there.
Pro
$0
- Chat on web, iOS, Android and desktop
- Artifact creation, editing and downloading
- Multi-file upload
- Unlimited custom agents and workflows
- All connectors (MES, ERP, QMS, SCADA, IoT and more)
- Triggers (Webhook, MQTT, Schedules)
- Messaging (Email, SMS, Slack, Teams and more)
- Pre-built agents and workflows matched to your role
- Generous usage limits
- In-product chat and self-service support
Team
$100
/user/month, billed annually- Everything in Pro, plus:
- Share artifacts, agents, workflows and connectors with your team
- Invite team members with role-based permissions
- Team-wide management with version control
- Higher usage limits
- Pre-built agents and workflows for teams
- Agent and email support
Site
$200
/user/month, billed annually- Everything in Team, plus:
- Create and manage groups
- Share and manage per group or entire org
- SSO and SCIM
- Activity and audit logs
- Higher usage limits
- Pre-built agents and workflows for sites
- 24/7 phone support
Enterprise
Volume
- Everything in Site, plus:
- Multi-site support
- On-prem A1 connector for integrations
- PrivateLink connectivity
- Pre-built cross-team workflows
- Dedicated delivery lead
- Dedicated support
Pre-built Agents & Workflows ensure A1 can execute a repeatable, consistent and compliant workflow every time.
You can't afford to let AI make up a new workflow every time it runs. A1's Agent and Workflow capability allows you to define the exact steps and actions A1 should take every time it runs.
A1 creates production-ready artifacts — CAPAs, deviations, reports and more.
A1 doesn't just answer questions. It generates real manufacturing documents — from CAPA reports to process summaries — that you can review, edit, and share with your team.
A1 is not a chatbot. It proactively responds 24/7 to alarms, events, schedules, file changes and more.
Production doesn't wait for a prompt. A1 can be configured to proactively respond to 'triggers' so you don't have to constantly be in front of your computer - let A1 handle the noise so you can focus on what's important.
One Agent. Every System.
A1 Agents and Workflows execute across your entire manufacturing tech stack — ERP, MES, QMS, EAM, SCADA, Historian, PLCs, IoT, and flat files. Connect point-to-point or through a middleware with Webhooks, MQTT or OPC-UA.
A1 can communicate with you where you are - Email, SMS, WhatsApp, Slack, Teams, and more
When an issue happens on the floor, the last thing you want is to have to log in to an app. A1 can reach out to you on the channels you already use so you don't lose a second.
Built on the same infrastructure that has been powering our Manufacturing Suite for 12 years.
We've been deploying cloud-native manufacturing software for over a decade in the most difficult and regulated industries in the world. Designed to be fault-tolerant, geographically redundant, and always available, you can have the confidence to use A1 on your most critical workflows.
Free for individuals. Scale to your team, site and enterprise.
Use A1 for free to automate your manual work. When you're ready to scale, pick a plan that fits your operations and grow from there.
Pro
$0
- Chat on web, iOS, Android & desktop
- Artifacts & multi-file upload
- Unlimited agents, workflows & connectors
- Triggers, messaging & pre-built agents for your role
- In-product chat & self-service support
Team
$100
/user/month, billed annually- Everything in Pro, plus:
- Share artifacts, agents, workflows & connectors
- Invite team members with roles & version control
- Higher usage; agent & email support
Site
$200
/user/month, billed annually- Everything in Team, plus:
- Groups, SSO & SCIM; per-group sharing
- Activity/audit logs; pre-built agents for sites
- 24/7 phone support
Enterprise
Volume
- Everything in Site, plus:
- Multi-site; on-prem connector & PrivateLink
- Cross-team workflows; dedicated delivery lead
- Dedicated support
Manufacturing Suite
Next-Gen Capabilities, Not a Patchwork of Point Solutions. Our Manufacturing Suite spans the full ISA-95 stack.
Schedule a Demo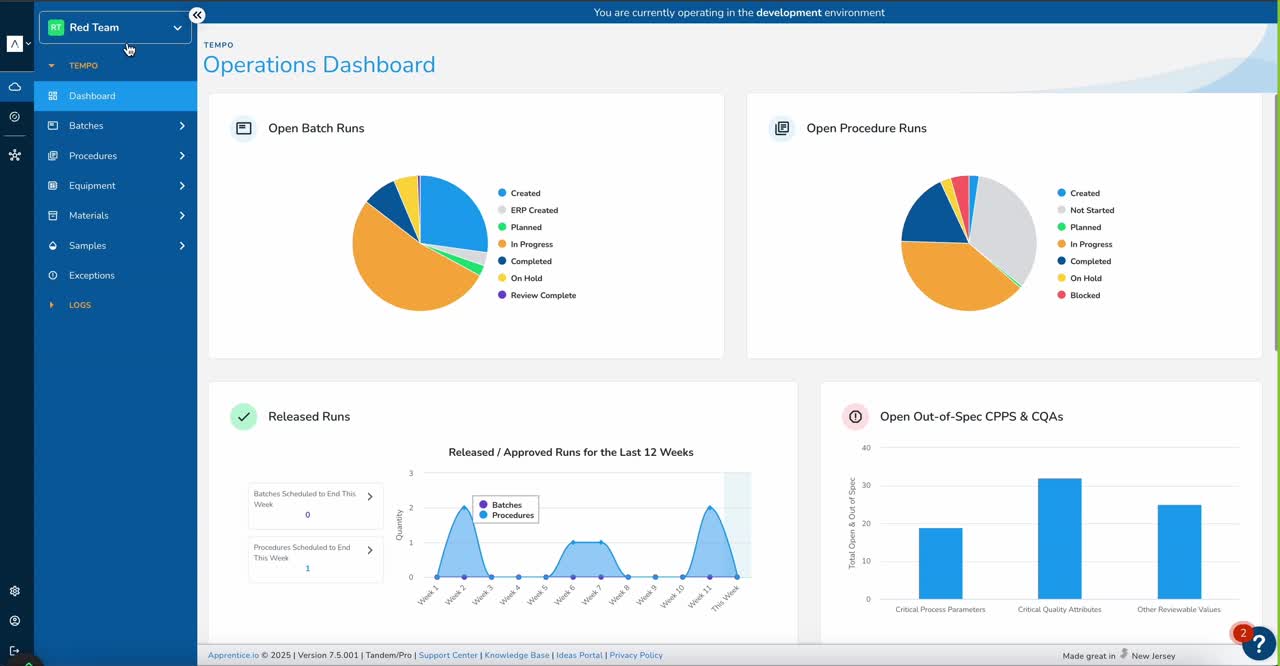
Connected Manufacturing Network
Unify teams, systems, and data across sites to streamline execution and deliver real-time insights across your enterprise.
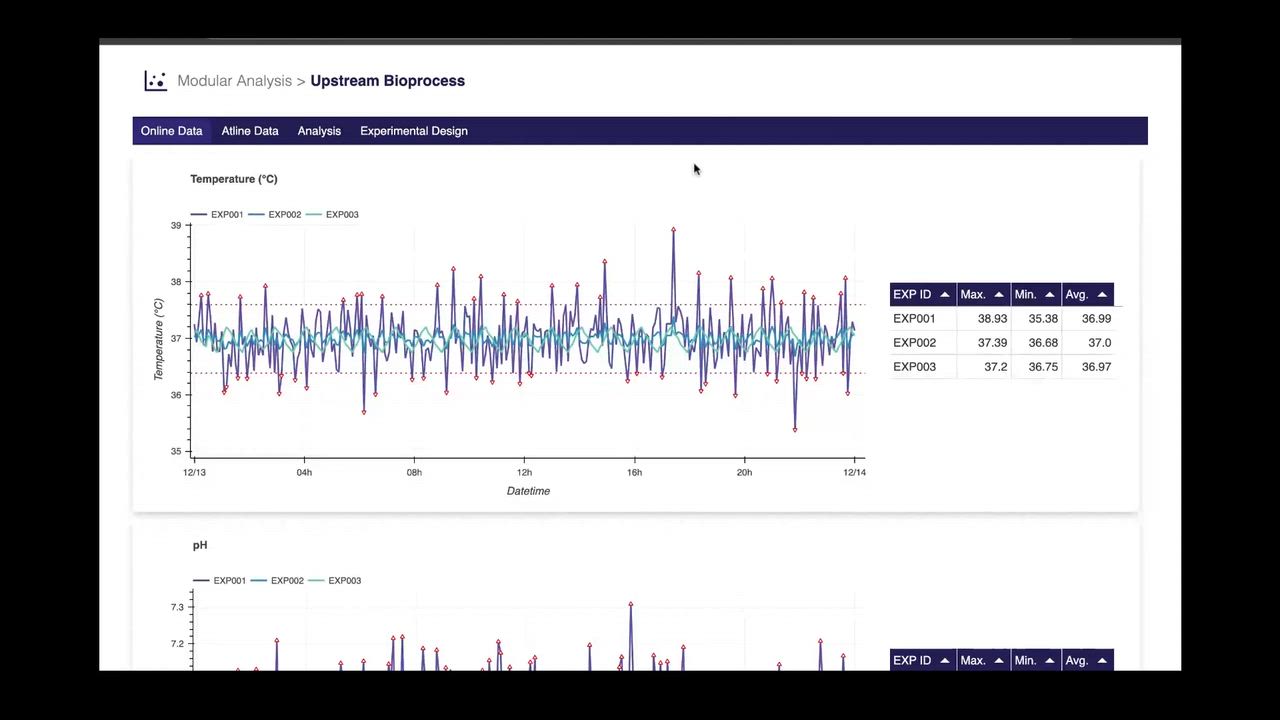
Manufacturing Intelligence
Unlock advanced intelligence and AI-driven analysis to predict risks, optimize processes, and enable smarter decisions at every stage of manufacturing and development.
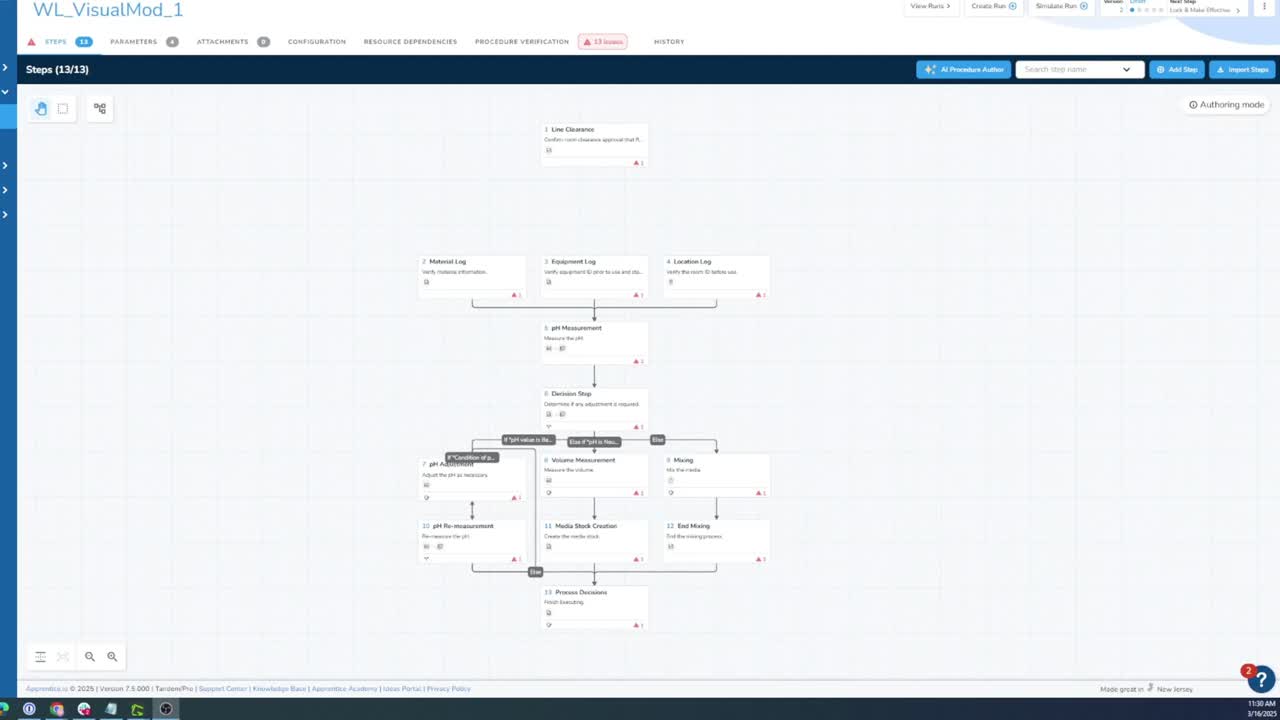
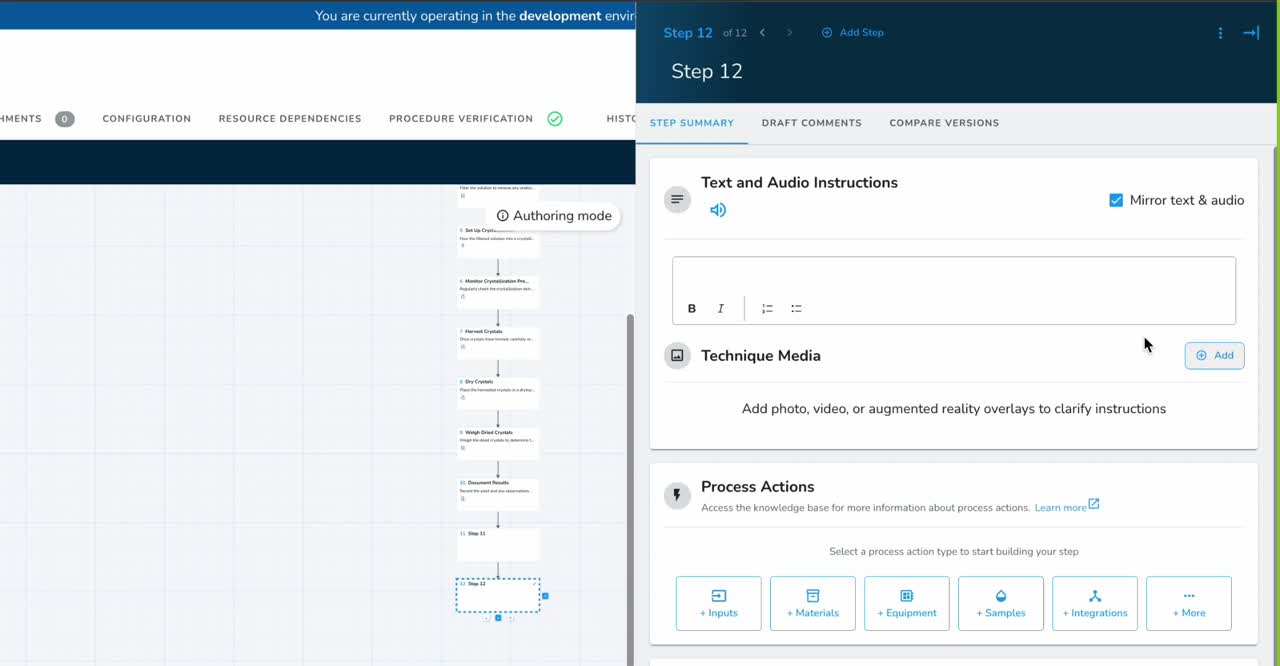
Manufacturing Execution System
Digitize and orchestrate your manufacturing — from recipe authoring to real-time production tracking — on one intuitive, no-code platform.
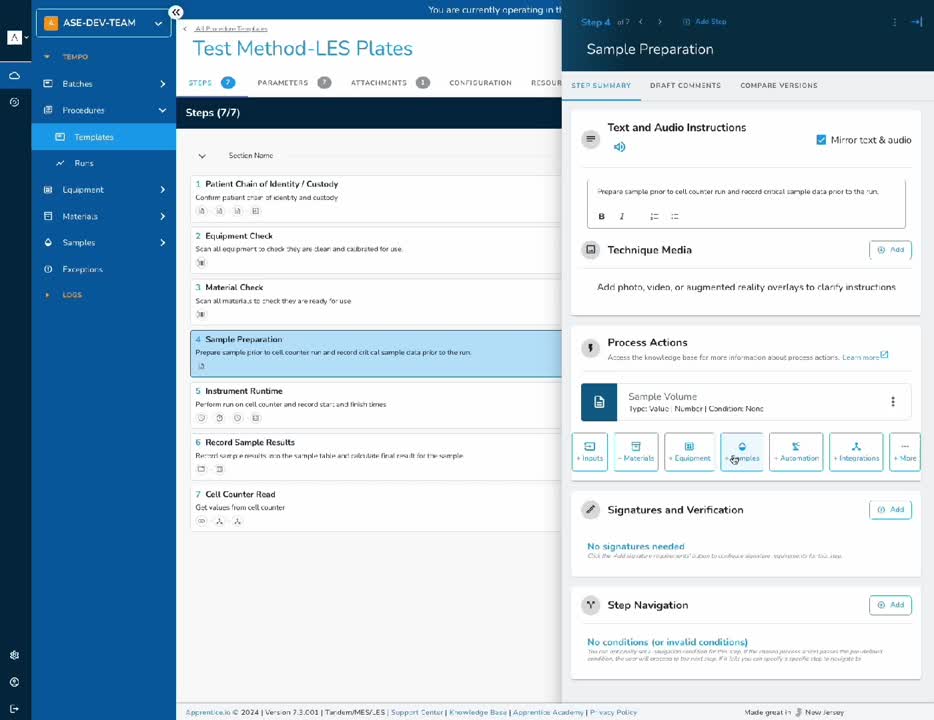
Laboratory Execution System
Speed up and scale your quality operations with digital procedures, sample tracking, and instrument management.
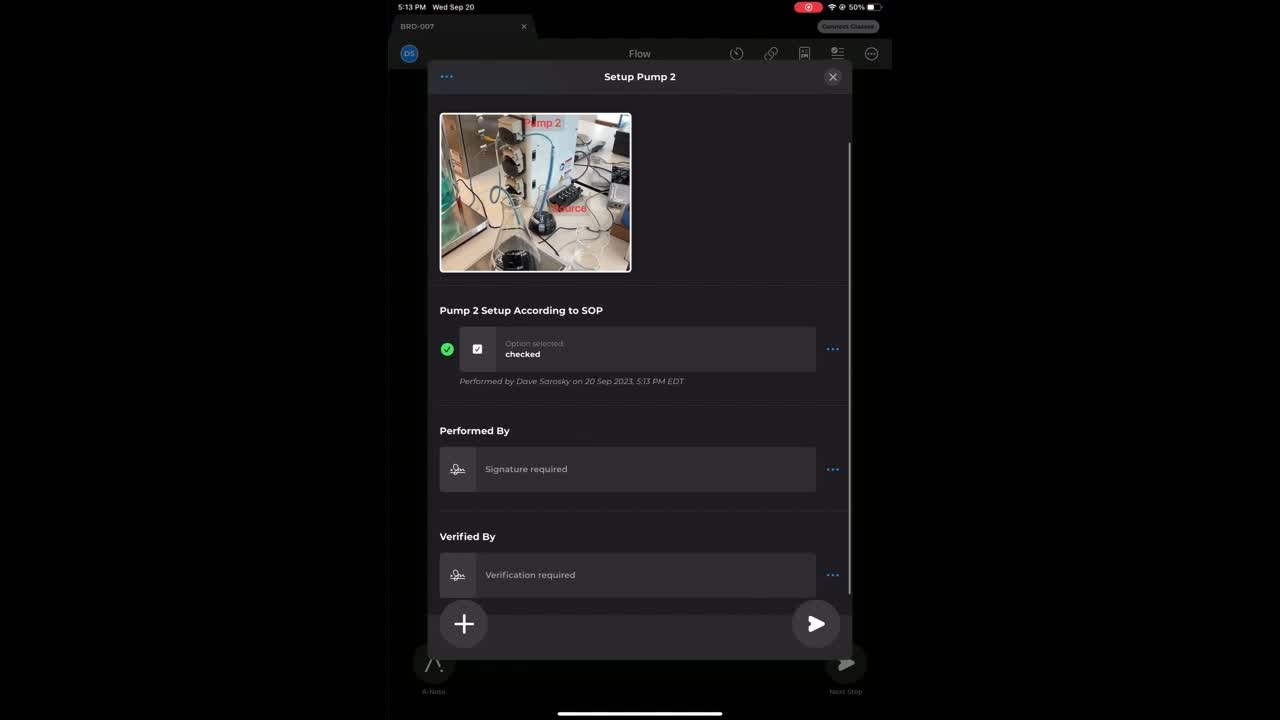
Edge Devices
Go paperless with dynamic, no-code instructions. Embed rich media like photos, videos, and AR overlays directly at the point of work.
Remote Assistance
Collaborate live with teams and partners via mobile, desktop, or AR headsets — anytime, anywhere.

Connected Manufacturing Network
Unify teams, systems, and data across sites to streamline execution and deliver real-time insights across your enterprise.

Manufacturing Intelligence
Unlock advanced intelligence and AI-driven analysis to predict risks, optimize processes, and enable smarter decisions at every stage of manufacturing and development.

Manufacturing Execution System
Digitize and orchestrate your manufacturing — from recipe authoring to real-time production tracking — on one intuitive, no-code platform.

Laboratory Execution System
Speed up and scale your quality operations with digital procedures, sample tracking, and instrument management.

Edge Devices
Go paperless with dynamic, no-code instructions. Embed rich media like photos, videos, and AR overlays directly at the point of work.

Remote Assistance
Collaborate live with teams and partners via mobile, desktop, or AR headsets — anytime, anywhere.

